METALS AND NON METALS
CLASS 10
ELEMENTS CAN BE CLASSIFIED AS METALS AND NON METALS ON TE BASIS OF THEIR PROPERTIES.
PHYSICAL PROPERTIES OF METALS AND NON METALS
Metals are malleable and ductile. .Non- metals are brittle (break easily). They are neither malleable nor ductile. Metals are good conductors of heat and electricity. Non-metals are bad conductors of heat and electricity. Except for graphite which is a good conduction of electricity. Metals are lustrous and can be polished. Non-metals are non-lustrous (dull) and cannot be polished. Except for iodine
Metals are solids at room temperature.Except for mercury which is a liquid metal. Non-metals may be solid, liquid or gases at the room temperature. Metals are strong and tough. Non-metals are not strong or tough.
- Carbon in the form of graphite is non-metal which conduct electricity.
- Iodine is non-metal which is lustrous having a shining surface.
- Carbon in the form of diamond is a non-metal which is extremely hard.
- Diamond is a non-metal which has a very high melting point and boiling point.
- sodium is the soft metal which can be cut by knife.
- Bromine is the only liquid non metal at room temperature.
- mercury (Hg) is the only liquid metal at room temperature.
Metal + Oxygen → Metal Oxide
Examples:reaction of Potassium with Oxygen: Potassium metal forms potassium oxide when reacts with oxygen.

Reaction of Sodium with Oxygen: Sodium metal forms sodium oxide when reacts with oxygen.
Lithium, potassium, sodium, etc. are known as Alkali-metals. Alkali metals react vigorously with oxygen.
Reaction of Copper metal with Oxygen: Copper does not react with oxygen at room temperature but when burnt in air, it gives oxide.
Silver, gold and platinum do not combine with the oxygen of air even at high temperature. They are the least reactive.
2. Reaction of metals with water: Metals form respective hydroxide or metal oxides and hydrogen gas when reacting with water.
Metal + Water → Metal hydroxide + Hydrogen
metal + steam=metal oxide + hydrogen
Most of the metals do not react with water. However, alkali metals react vigorously with water.
copper lead gold silver do not react with any form of water.
Reaction of Sodium metal with Water: Sodium metal forms sodium hydroxide and liberates hydrogen gas along with lot of heat when reacting with water.
Reaction of Calcium metal with Water: Calcium forms calcium hydroxide along with hydrogen gas and heat when react with water.
Reaction of Magnesium metal with Water: Magnesium metal reacts with hot water slowly and forms magnesium hydroxide and hydrogen gas.it never reacts with cold water.
Reaction of Aluminium metal with Water: Reaction of aluminium metal with cold water is too slow to come into notice. But when steam is passed over aluminium metal, aluminium oxide and hydrogen gas are produced.
2Al + 3H2O → Al2O3 + 2H2
Reaction of Zinc metal with Water: Zinc metal produces zinc oxide and hydrogen gas when steam is passed over it. Zinc does not react with cold water.
Reaction of Iron with Water: Reaction of iron with cold water is very slow and comes into notice after a long time. Iron forms rust (iron oxide) when reacts with moisture present in the atmosphere. Iron oxide and hydrogen gas are formed by passing of steam over iron metal.
Both calcium (Ca) and magnesium (Mg) are heavier than water but still float over it: Both calcium and magnesium float over water surface because hydrogen gas is evolved when these metals react with water. It is in the form of bubbles which stick on the metal surface. Therefore, they float over it.
Other metals usually do not react with water or react very slowly. Lead, copper, silver and gold do not react with steam. Thus, the order of reactivity of different metals towards water may be written as :
K > Na > Ca > Mg > Ae > Zn > Fe > Pb > Cu > Ag > Au
3. Reaction of metals with dilute acid:
Metals form respective salts when reacting with dilute acid.
Metal + dil. acid → Metal salt + Hydrogen
Reaction of Sodium metal with dilute hydrochloric acid: Sodium metal gives sodium chloride and hydrogen gas when react with dilute hydrochloric acid.
Reaction of Magnesium metal with dilute hydrochloric acid: Magnesium chloride and hydrogen gas are formed when magnesium reacts with dilute hydrochloric acid.
Reaction of Zinc with dilute sulphuric acid: Zinc sulphate and hydrogen gas are formed when zinc reacts with dilute sulphuric acid. This method is used in the laboratory to produce hydrogen gas.
Hydrogen (H2) gas is not evolved when metal is treated with nitric acid (HNO3):
Nitric acid is strong oxidising agent and it oxidises the hydrogen gas (H2) liberated into water (H2O) and itself get reduced to some oxide of nitrogen like nitrous oxide (N2O)3 nitric oxide (NO) and nitrogen dioxide (NO2).
Copper, gold, silver are known as noble metals. These do not react with water or dilute acids.
The order of reactivity of metal towards dilute hydrochloric acid or sulphuric acid is in the order;
K > Na > Ca > Mg > Al > Zn > Fe > Cu > Hg > Ag
Metal Oxides
Chemical Properties: Metal oxides are basic in nature. The aqueous solution of metal oxides turns red litmus blue.
Reaction of Metal oxides with Water: Most of the metal oxides are insoluble in water. Alkali metal oxides are soluble in water. Alkali metal oxides give strong base when dissolved in water.
Reaction of Sodium oxide with Water: Sodium oxide gives sodium hydroxide when reacts with water.
Reaction of Potassium oxide with Water: Potassium oxide gives potassium hydroxide when reacts with water.
Reaction of Zinc oxide and Aluminium oxide: Aluminium oxide and zinc oxide are insoluble in water. Aluminium oxide and zinc oxide are amphoteric in nature. An amphoteric substance shows both acidic and basic characters. It reacts with base like acid and reacts with an acid like a base.
When zinc oxide reacts with sodium hydroxide, it behaves like an acid. In this reaction, sodium zincate and water are formed.
Zinc oxide behaves like a base when reacts with acid. Zinc oxide gives zinc chloride and water on reaction with hydrochloric acid.
In a similar way, aluminium oxide behaves like a base when reacts with acid and behaves like acid when reacts with a base.
Aluminium oxide gives sodium aluminate along with water when reacts with sodium hydroxide.
Aluminium oxide gives aluminium chloride along with water when it reacts with hydrochloric acid.
Reactivity Series of Metals: The order of intensity or reactivity of metal is known as Reactivity Series. Reactivity of elements decreases on moving from top to bottom in the given reactivity series.
In the reactivity series, copper, gold, and silver are at the bottom and hence, least reactive. These metals are known as Noble metals. Potassium is at the top of the series and hence, most reactive.
Reactivity of some metals are given in descending order :
K > Na > Ca > Mg > Al > Zn > Fe > Pb > Cu
4. Reaction of metals with solution of other metal salts: Reaction of metals with the solution of other metal salt is displacement reaction. In this reaction, more reactive metal displaces the less reactive metal from its salt.
Metal A + Salt of metal B → Salt of metal A + Metal B
Examples :
Iron displaces copper from copper sulphate solution.
Similarly, aluminium and zinc displace copper from the solution of copper sulphate.
In all the above examples, iron, aluminium and zinc are more reactive than copper. This is why they displace copper from its salt solution.
When copper is dipped in the solution of silver nitrate, it displaces silver and forms copper nitrate.
In the reaction, copper is more reactive than silver and hence, displaces silver from silver nitrate solution.
Silver metal does not react with copper sulphate solution because silver is less reactive than copper and not able to displace copper from its salt solution.
Similarly, when gold is dipped in the solution of copper nitrate, no reaction takes place because copper is more reactive than gold.
In similar way, no reaction takes place when copper is dipped in the solution of aluminium nitrate because copper is less reactive than aluminium.
Chemical properties of Non-metals
1. Reaction of Non-metals with Oxygen: Non-metals form respective oxide when reacting with oxygen.
Non-metal + Oxygen → Non-metallic oxide
When carbon reacts with oxygen, carbon dioxide is formed along with the production of heat.
When carbon is burnt in an insufficient supply of air, it forms carbon monoxide. Carbon monoxide is a toxic substance. Inhaling of carbon monoxide may prove fatal.![]()
Sulphur gives sulphur dioxide when reacting with oxygen. Sulphur catches fire when exposed to air.![]()
When hydrogen reacts with oxygen it gives water.![]()
Non-metallic Oxide: Non-metallic oxides are acidic in nature. The solution of non-metal oxides turns blue litmus red.
Carbon dioxide gives carbonic acid when dissolved in water.![]()
Sulphur dioxide gives sulphurous acid when dissolved in water.
Sulphur dioxide gives sulphuric acid when reacts with oxygen.
2. Reaction of Non-metal with Chlorine: Non-metal gives respective chloride when they react with chlorine gas.
Non-metal + Chlorine → Non-metal chloride
Hydrogen gives hydrogen chloride and phosphorous gives phosphorous trichloride when reacting with chlorine.
3. Reaction of Non-metals with Hydrogen: Non-metals reactive with hydrogen to form covalent hydrides.
Non-metal + Hydrogen → Covalent Hydride
Sulphur combines with hydrogen to form a covalent hydride is called Hydrogen sulphide.
Nitrogen combines with hydrogen in presence of an iron catalyst to form covalent hydride ammonia.
Non-metals do not react with water (or steam) to evolve Hydrogen gas.
Non-metals do not react with dilute acids.
4. Reaction of Metal and Non-metal: Many metals form ionic bonds when they react with non-metals. Compounds so formed are known as Ionic Compounds.
Ions: Positive or negative charged atoms are known as ions. Ions are formed because of loss or gain of electrons. Atoms form ions obtain by the electronic configuration of the nearest noble gas.
Positive ion: A positive ion is formed because of the loss of electrons by an atom.
Following are some examples of positive ions:
Sodium forms sodium ion because of the loss of one electron. Because of the loss of one electron, one positive charge comes over sodium.![]()
Magnesium forms positive ion because of the loss of two electrons. Two positive charges come over magnesium because of loss of two electrons.
Negative ion: A negative ion is formed because of the gain of an electron.
Some examples are given below :
Chlorine gains one electron in order to achieve a stable configuration. After the loss of one electron, chlorine gets one negative charge over it forming chlorine ion.
Ionic Bonds: Ionic bonds are formed because of transfer of electrons from metal to non¬metal. In this course, metals get positive charge because of transfer of electrons and non-metal gets negative charge because of acceptance of electrons. In other words, bond formed between positive and negative ion is called Ionic Bond.
Since, a compound is electrically neutral, so to form an ionic compound, negative and positive both ions must be combined.
Some examples are given below:
Formation of Sodium Chloride (NaCl): In sodium chloride, sodium is a metal (alkali metal) and chlorine is a non-metal.
Atomic number of sodium = 11
Electronic configuration of sodium : 2, 8, 1
Number of electrons in outermost orbit = 1
Valence electrons = Electrons in outermost orbit = 1
Atomic number of chlorine = 17
Electronic configuration of chlorine : 2, 8, 7
Electrons in outermost orbit = 7
Therefore, valence electrons = ?
Sodium has one valence electron and chlorine has seven valence electrons. Sodium requires losing one electron to obtain stable configuration and chlorine requires gaining one electron in order to obtain stable electronic configuration. Thus, in order to obtain stable configuration, sodium transfers one electron to chlorine. After loss of one electron, sodium gets one positive charge (+) and chlorine gets one negative charge after gain of one electron. Sodium chloride is formed because of transfer of electrons. Thus, ionic bond is formed between sodium and chlorine. Since, sodium chloride is formed because of ionic bond, thus, it is called Ionic compound. In similar way, potassium chloride (KCl) is formed.
Properties of Ionic compound
- Ionic compounds are solid. Ionic bond has a greater force of attraction because of which ions attract each other strongly. This makes ionic compounds solid.
- Ionic compounds are brittle.
- Ionic compounds have high melting and boiling points because force of attraction between ions of ionic compounds is very strong.
- Ionic compounds generally dissolve in water.
- Ionic compounds are generally insoluble in organic solvents; like kerosene, petrol, etc.
- Ionic compounds do not conduct electricity in the solid state.
- The solution of ionic compounds in water conduct electricity. This happens because ions present in the solution of ionic compound facilitate the passage of electricity by moving towards opposite electrodes.
- Ionic compounds conduct electricity in the molten state.
Access Answers to NCERT Class 10 Science Chapter 3 – Metals and Non-metals ( All In text and Exercise Questions Solved)
In-text questions set 1 Page number 40
1. Give an example of a metal which
(i) Is a liquid at room temperature?
(ii) Can be easily cut with a knife?
(iii) Is the best conductor of heat?
(iv) Is a poor conductor of heat?
Solution:
(i) Mercury is the metal which is liquid at room temperature
(ii) Sodium and potassium are the metals which can be cut with a knife
(iii) Silver is the best conductor of heat
(iv) Mercury and lead are poor conductor of heat.
2. Explain the meanings of malleable and ductile.
Solution:
- Metals which can be beaten to sheets are said to be malleable
- Metals which can be drawn into thin wires are said to be ductile
In-text questions set 2 Page number 46
1. Why is sodium kept immersed in kerosene oil?
Solution: Sodium is a reactive metals, if kept open it will react with oxygen to explore and catch fire. Sodium metal is kept immersed in kerosene to prevent their reaction with oxygen, moisture and carbon dioxide of air.
2. Write equations for the reactions of
(i) iron with steam
(ii) calcium and potassium with water
Solution: (i) Iron reacts with steam to form a magnetic oxide of Fe with the liberation of H2.
3Fe(s) + 4H2O(g) → Fe3O4(s) + 4H2(g)
(ii) Calcium reacts with water to form calcium hydroxide and hydrogen.
Ca(s) + 2H2O(I) → Ca(OH)2(aq) + H2(g)
Potassium reacts with cold water violently immediately with evolution of H2 which catches fire.
2K(s) + 2H2O(I) → 2KOH(aq) + 2H2(g)
3. Samples of four metals A, B, C and D were taken and added to the following solution one by one. The results obtained have been tabulated as follows
| Metal | Iron(II) sulphate | Copper(II) sulphate | Zinc sulphate | Silver Nitrate |
| A | No reaction | Displacement | – | – |
| B | Displacement | – | – | – |
| C | No reaction | No reaction | No reaction | Displacement |
| D | No reaction | No reaction | No reaction | No reaction |
Use the Table above to answer the following questions about metals A, B, C and D.
- Which is the most reactive metal?
- What would you observe if B is added to a solution of Copper (II) sulphate?
- Arrange the metals A, B, C and D in the order of decreasing reactivity.
Solution:
(i) Metal B is the most reactive as it gives displacement reaction with iron (II) sulphate.
(ii) When metal B is added to copper (II) sulphate solution, a displacement reaction will take place because of which the blue colour of copper (II) sulphate solution will fade and a red-brown deposit of copper will be formed on metal B.
(iii)Metal B is the most reactive because it displaces iron from its salt solution. Metal A is less reactive because it displaces copper from its salt solution. Metal C is still less reactive because it can displace only silver from its salt solution and metal D is the least reactive because it cannot displace any metal from its salt solution. Hence, the decreasing order of reactivity of the metals is B > A > C > D.
4. Which gas is produced when dilute hydrochloric acid is added to a reactive metal? Write the chemical reaction when iron reacts with dilute H2SO4.
Solution: Hydrogen gas is liberated when dilute HCl is added to a reactive metal.
Fe(s) + H2SO4(aq) → FeSO4(aq) + H2(g)
5. What would you observe when zinc is added to a solution of iron (II) sulphate? Write the chemical reaction that takes place.
Solution: Zinc is more reactive (more electro positive) than iron. Therefore Zinc displaces Iron from its salt solution. The colour of ferrous sulphate is pale green, which turns colourless.
FeSO4 + Zn → ZnSO4 + Fe(s)
Light green Zinc sulphate(Colourless)
In-text questions set 3 Page number 49
1. (i) Write the electron-dot structures for sodium and oxygen.
(ii) Show the formation of Na2O and MgO by the transfer of electrons.
(iii)What are the ions present in these compounds?
Solution: (i) Sodium:

Oxygen:

(ii) Formation of Magnesium oxide:
When magnesium reacts with oxygen, the magnesium atom transfers its two outermost electrons to an oxygen atom. By losing two electrons, the magnesium atoms form a magnesium ion (Mg2+) and by gaining two electrons, the oxygen atom forms an oxide ion (O2-).
Mg: + → MgO
→ MgO
Formation of Sodium oxide:
Two sodium atoms transfer their 2 outermost electrons to an oxygen atom. By losing two electrons, the two sodium atoms form sodium ions (2Na+). And by gaining two electrons, the oxygen atom forms an oxide ion (O2-).
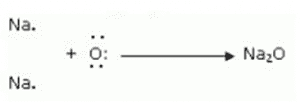
(iii) The ions present in sodium oxide compound (Na2O) are sodium ions (2Na+) and oxide ions (O2-).
The ions present in Magnesium oxide compound (MgO) are magnesium ions Mg2+ and oxide ions (O2-).
2. Why do ionic compounds have high melting points?
Solution: Ionic compounds are the ones which has both positive and negative charges. Hence there will be strong force of attraction between them. This make expenditure of lot of heat to break this force of attraction hence ionic compounds have high melting points.
In-text questions set 4 Page number 53
1. Define the following terms.
(i) Mineral
(ii) Ore
(iii) Gangue
Solution:
- Minerals are compounds (also known as elements) which are found naturally in the earth’s crust. E.g. Alums, K2SO4.Al2(SO4)3.24H2O, etc.
- Ores are minerals from which metal can be extracted Ex: Bauxite Al2O3.2H2O is the ore of Al, copper pyrite CuFeS2.All minerals are not considered as ores but all ores are also minerals.
- Ores mined from the earth are naturally contaminated with sand, rocky materials. There are impurities present in the ore which are known as gangue.
2. Name two metals which are found in nature in the free state
Solution: Gold and platinum are the two metals found in Free State in nature.
3. What chemical process is used for obtaining a metal from its oxide?
Reduction method is used to obtain metal from its oxide. Ex: Zinc oxide is reduced to metallic zinc by Heating with carbon.
ZnO + C → Zn + CO
Ex: Lead oxide is reduced to lead by heating with carbon
PbO +C → Pb + CO
In-text questions set 5 Page number 55
1. Metallic oxides of zinc, magnesium and copper were heated with the following metals.
| Metal | Zinc | Magnesium | Copper |
| Zinc Oxide | |||
| Magnesium Oxide | |||
| Copper Oxide |
Solution:
A more reactive metal can displace a less reactive metal from its oxide. Among Zinc, Magnesium, and Copper metals, magnesium is the most reactive, copper is the least reactive metal and zinc is less reactive .The displacement reaction will take place in the following cases
| Metal | Zinc | Magnesium | Copper |
| Zinc Oxide | – | Displacement | – |
| Magnesium Oxide | – | – | – |
| Copper Oxide | Displacement | Displacement | – |
2. Which metals do not corrode easily?
Solution: Gold and platinum are the metals which do not corrode easily
3. What are alloys?
Solution: An alloy is a homogeneous mixture of two or more metals, or a metal and a non-metal.
Exercise questions Page number 56-57
1. Which of the following pairs will give displacement reactions?
(a) NaCl solution and copper metal
(b) MgCl2 solution and aluminium metal
(c) FeSO4 solution and silver metal
(d) AgNO3 solution and copper metal
Solution: Option d i.e AgNO3 solution and copper is correct answer. Copper displace the silver cations (reducing them to the elemental metal), in the process copper itself being oxidised to Copper II cations (Cu2+) and going into solution. So silver metal precipitating out and a copper II nitrate solution will be remaining.
Cu(s) + 2AgNO3 (aq) → Cu(NO3)2 (aq) + 2Ag (s)
2. Which of the following methods is suitable for preventing an iron frying pan from rusting?
- Applying grease
- Applying paint
- Applying a coating of zinc
- All of the above
Solution: Answer is (c) Applying a coat of Zinc
Though applying grease and applying paint prevents iron from rusting but we cannot apply these methods on frying pan hence applying a coat of Zinc is most appropriate method to prevent an iron pan from rusting.
3. An element reacts with oxygen to give a compound with a high melting point. This compound is also soluble in water. The element is likely to be
(a) Calcium
(b) Carbon
(c) Silicon
(d) Iron
Solution: Correct answer is option (a) i.e Calcium.
Calcium reacts with oxygen to give calcium oxide. Calcium oxide is soluble in water to give Calcium Hydroxide.
Carbon forms carbon-oxide with oxygen which is gas hence option B is wrong
Silicon reacts with oxygen and forms silicon dioxide. This is insoluble in water. So option C is not correct.
Iron reacts with oxygen and forms Iron dioxide. This is insoluble in water. So option D is not correct.
4. Food cans are coated with tin and not with zinc because
(a) Zinc is costlier than tin.
(b) Zinc has a higher melting point than tin.
(c) Zinc is more reactive than tin.
(d) Zinc is less reactive than tin.
Solution: Answer is c. Food cans are coated with tin and not with zinc because Zinc is more reactive that is electro positive than tin.
5. You are given a hammer, a battery, a bulb, wires and a switch.
(a) How could you use them to distinguish between samples of metals and non-metals?
(b) Assess the usefulness of these tests in distinguishing between metals and non-metals.
Solution:
- Metals are malleable and can be easily drown into sheets by hitting with hammer. On the other hand if we beat non-metals they break down and they cannot be drawn into sheets as they are non-malleable. Metals of good conductors of electricity hence they make bulb when you connect metals with a battery, wire and bulb. Similarly If non-metals are bad conductors of electricity chance they fail to lit up the bulb on connecting with wire and battery.
- These experiments can be helpful to demonstrate the malleability and electric conductivity of the metals and non-metals
6. What are amphoteric oxides? Give two examples of amphoteric oxides
Solution: Oxides that react with both acids and bases to form salt and water are known as amphoteric oxides. Examples: PbO and Al2O3.
Amphoteric oxides are the one which reacts with both acids and bases to form salt and water. Examples: Lead oxide – PbO and Aluminium oxide – Al2O3.
7. Name two metals which will displace hydrogen from dilute acids, and two metals which will not.
Solution: Zinc (Zn) and Magnesium (Mg) are the two metals which will displace Hydrogen from dilute acids as they are very reactive metals. Gold (Au) and Silver (Ag) are the metals which will not replace Hydrogen from dilute acids as these metals are less reactive.
8. In the electrolytic refining of a metal M, what would you take as the anode, the cathode and the electrolyte?
Solution: In the process of electrolytic refining of metal called ‘M’, An impure and thick block of metal M. is considered as anode, Thin strip or wire of pure metal M is taken as cathode. A suitable salt solution of metal M is considered as the electrolyte.
9. Pratyush took sulphur powder on a spatula and heated it. He collected the gas evolved by inverting a test tube over it, as shown in figure below.
(a) What will be the action of gas on
(i) dry litmus paper?
(ii) moist litmus paper?
(b) Write a balanced chemical equation for the reaction taking place.
Solution: a) When sulphur powder is burnt in the air sulphur-di-oxide is formed.
(i) Sulphur-di-oxide does not have any effect on dry litmus paper.
(ii) Sulphur-di-oxide turn the moist litmus paper from blue to red as contact of SO2 with water turns to sulfurous acid.
(b) S(s) + O2(g) → SO2(g)
SO2(g) + H2O →H2SO3
10. State two ways to prevent the rusting of iron.
Solution:
- Iron can be prevented from rusting by coating the surface of the iron with rust proof paints
- By applying Oil/grease on the surface of iron objects as it will prevent the iron surface to get in contact with air consisting of moisture.
11. What type of oxides are formed when non-metals combine with oxygen?
Solution: When non-metals combine with oxygen it forms either acidic or neutral oxides. Ex: N2O5 or N2O3 is an acidic oxide; CO is a neutral oxide.
12. Give reasons
(a) Platinum, gold and silver are used to make jewellery.
(b) Sodium, potassium and lithium are stored under oil.
(c) Aluminium is a highly reactive metal, yet it is used to make utensils for cooking.
(d) Carbonate and sulphide ores are usually converted into oxides during the process of extraction
Solution:
(a) Platinum, gold and silver are used to make jewellery for these metals are very less reactive hence they are not affected by air, water or most chemicals. These metals have a lot of luster and they are malleable and ductile in nature and also high corrosion resistance in nature.
(b) Sodium, potassium and lithium readily react with water to produce a lot of heat. As a result, Hydrogen evolved in the reaction results in a fire. On exposure to water they react with moisture (water droplets) present in the atmosphere, In order to prevent contact with water hence these metals are stored under oil.
(c) Aluminium forms on its surface a nonreactive surface of aluminium oxide. Such coating prevents other compounds from reacting to aluminium. So aluminium is being used to produce utensils for cooking.
(d) Reducing metal oxide into free metal is easy. Additionally, because it is easier to obtain metals directly from their oxides than from their carbonates or sulphides, the carbonate and sulphide ores are first transformed to oxides to obtain the metals.
13. You must have seen tarnished copper vessels being cleaned with lemon or tamarind juice. Explain why these sour substances are effective in cleaning the vessels.
Solution: Tarnished copper vessels being cleaned with lemon or tamarind because this sour substance contains acids which dissolve the coating of copper oxide or basic copper carbonate present on the surface or tarnished copper vessels. This makes them shining red-brown again. Hence they are very effective in cleaning tarnished copper vessels.
14. Differentiate between metal and non-metal on the basis of their chemical properties.
Solution:
| Metals | Non-metals |
| When metals are heated with oxygen, they form ionic oxides which are basic in nature and form bases on dissolving with water. This turn red litmus paper to blue. | When non-Metals are heated with oxygen, they form covalent oxides which are acidic in nature which form acid on dissolving with water. This turn blue litmus paper to red. |
| They are electro positive, lose electrons readily and become a positive ion. | They are electro negative, gain electrons and become negative ions. |
| Metals are lustrous. | Non-metals are non-lustrous; graphite is the exception |
| Reducing agents. | Good oxidizing agents. |
| Metals are the good conductors of electricity and heat. | Non-metals are non-conductors of electricity and heat; graphite is the exception |
| All metals are solids except mercury. | Non-metals are in solid-liquid and gaseous states |
15. A man went door to door posing as a goldsmith. He promised to bring back the glitter of old and dull gold ornaments. An unsuspecting lady gave a set of gold bangles to him which he dipped in a particular solution. The bangles sparkled like new but their weight was reduced drastically. The lady was upset but after a futile argument the man beat a hasty retreat. Can you play the detective to find out the nature of the solution he had used?
Solution: Goldsmith used the solution called Aqua regia which is called as royal water in Latin. It is the mixture of concentrated Hydrochloric acid and concentrated nitric acid in the ratio of 3:1. Aqua regia is capable of dissolving noble metals like gold and platinum. When upper-layer of dull gold ornament is dissolved they lose their weight.
16. Give reasons why copper is used to make hot water tanks and not steel (an alloy of iron).
Solution: Copper is used to make hot water tanks and not steel (an alloy of iron) because copper does not reacts with either water or steam whereas iron reacts with steams to corrode the tank.
What are Metalloids?
Metalloids can be defined as chemical elements whose physical and chemical properties fall in between the metal and the non metal categories. Boron, germanium, silicon, antimony, arsenic, and tellurium are the six most widely recognized metalloids. Apart from these elements, the following elements are also known to be classified as metalloids in some circumstances:
- Carbon
- Aluminium
- Selenium
- Polonium
- Astatin
It can be noted that all eleven of these elements can be found on the regular periodic table in a diagonal region of the p block which extends from boron (which is placed at the upper left) to astatine (which is placed at the lower right). Some periodic tables have a dividing line between metals and nonmetals, and below this line, the metalloids can be found.
Typically, metalloids have metallic appearances but they are usually brittle and only mediocre electricity conductors. Chemically, these elements usually behave as non-metals. Metalloids have the ability to form metallic alloys. Other physical properties and chemical properties of the metalloid elements are usually intermediate in nature. In general, these elements are extremely fragile and, therefore, do not have many structural applications. In alloys, catalysts, biological agents, glasses, flame retardants, optical storage and optoelectronics, semiconductors, pyrotechnics, and electronics, Metalloids and their compounds are used.
General Properties of Metalloids
- Metalloids typically look like metals. However, these elements often behave like non-metals.
- Physically, metalloids are brittle, somewhat shiny substances that are usually solid at ambient temperatures.
- These elements usually have intermediate to fairly strong electrical conductivity
- Metalloids are known to have electronic band structures that are similar to semimetals or semiconductors.
- Chemically, these elements usually act as non-metals (in a relatively weak manner)
- These elements generally have intermediate energies of ionization and values of electronegativity
- Metalloids are known to form amphoteric or weakly acidic oxides.
- These elements have the ability to form metallic alloys.
- Many of the other physical and chemical properties of metalloids, in essence, are intermediate.




No comments:
Post a Comment